1. What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
2. Write the IUPAC name of the following organic compound:
C(CH3)3
CH3-CH2- CH2-CHCH(Cl)CH2CH3
3. Arrange the following in decreasing order of basic strength in gas phase:
C2H5NH2, (C2H5)2NH,(C2H5)3N , NH3
4. Write the structure of the following:
4-fluoro-2-hydroxyacetophenone
5. Name the reagent used in the conversion of :
i) Butan-2-one to butan-2-ol
ii) Bromination of phenol to 2,4,6- tribromophenol
6. i) On mixing acetone with chloroform, a reduction occurs in total volume. What type of deviations from ideal behavior for solutions is shown in this case and why?
ii) How can the direction of osmosis be reversed? Write one use of reverse osmosis.
7. Explain the following observations:
i) Transition metals generally form coloured compounds.
ii) The highest oxidation state of a metal is exhibited in its oxide or fluoride.
OR
What happens when KI solution is added to
i) Alkaline solution of KMnO4?
ii) Acidified solution of KMnO4?
8. Explain the following terms:
i) Electrophoresis
ii) Dialysis
9. For a certain reaction, variation in the concentration, ln[R] Vs time(s) plot is given below:
i) What is the order of the reaction?
ii) What are the units of rate constant k?
iii) Give the relationship between k and t1/2?
iv) What does the slope of the above line indicate?
10. i) Why is it necessary to remove CO when ammonia is obtained by Haber’s process?
ii) Explain what is observed when a beam of light is passed through a colloidal solution?
11. The density of chromium metal is 7.2 g cm-3. If the unit cell has edge length of 289 pm, determine
i) The type of unit cell
ii) The radius of chromium metal
(At.mass of Cr = 52 u, N0= 6.023 x 1023 )
12. i) Write the steps involved in the preparation of K2Cr2O7 from Na2CrO4.
ii) What is the effect of increasing pH on a solution of dichromate?
13. Explain why
i) Benzylic and allylic halides follow SN1 mechanism?
ii) Iodination of alkanes require the presence of an oxidizing agent like HNO3 or HIO3.
iii) Grignard reagent should be prepared under anhydrous conditions.
14. Assuming complete ionization, calculate the expected freezing point of solution prepared by dissolving 7.00g of Glauber’s salt, Na2SO4.10H2O in 100 g of water.
(Kf for water = 1.86 K kg mol-1)
(RAM of Na = 23, S = 32 , O = 16, H =1 u)
15. How are the following conversions carried out?
i) Ethanoic acid to methanamine
ii) Nitromethane to dimethyl amine
iii) Chlorobenzene to p-chloroaniline
16. i) Name the method used for refining of zirconium.
ii) What is the role of CO in the extraction of iron?
iii) What is meant by the term pyrometallurgy?
17. Write the mechanism of the following reaction :
H+
2CH3CH2OH → CH3CH2-O-CH2CH3
18. A first order reaction takes 40 minutes for 30% decomposition. Calculate t1/2 for this reaction.
OR
Consider the reaction : 2A + B → C + D
Following results were obtained in experiments designed to study the rate of reaction:
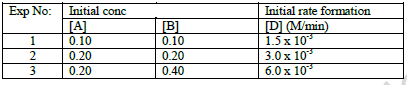
i) Write the rate law for the reaction.
ii) Calculate the value for the rate constant for the reaction.
19. i) What type of linkage holds together the monomers of DNA?
ii) What are the expected products of hydrolysis of lactose?
iii) What happens when D-glucose is treated with
a) HI
b) HNO3
20. i) Write the formula for the following coordination compound:
Amminebromidochloridonitrito-N-platinate(II)
ii) Why is geometrical isomerism not possible in tetrahedral complexes having two different types of unidentate ligands coordinated with the central metal ion?
iii) On the basis of crystal field theory, write the electronic configuration of d4 ion if Δt> p.
21. Assign reasons for the following :
i) H2S is more acidic than H2O.
ii) NH3 is more basic than PH3.
iii) Sulphur has a greater tendency for catenation than oxygen.
22. Write the names of the monomers of the following polymers :
i) Nylon-6,6
ii) Bakelite
iii) Terelene
Please click on below link to download CBSE Class 12 Chemistry Sample Paper 2022 Set A Solved

